Sometimes we do procedures to just get it over with and do it mechanically without fully understanding why it really works. Thus, when it doesn’t we blame it all to the materials or every one else except ourselves. Learning how our materials work gives us a better understanding how we can really do it better and be able to choose the best material to do the job at hand. Have you ever really wonder why silane is needed when we do composite or porcelain repair? Or, why we need it for cementation for veneers or all porcelain or no-metal indirect restorations? Well, it is all because silane is part of the coupling and components of what makes our composite resins.
In the current world of restoration that doesn’t involve extension for prevention and undercuts as a necessity to hold our restorations in place because adhesion for retention is the name of the game, then, it is critical we know our materials’ compositions. The principles of composite resin to stay in place relies heavily on how we can properly bond our composite resins to the tooth surface. We all know that bonding on the enamel is easier , thus, we can’t hardly botch that part but the reason why composite resin restorations become technique sensitive material is due to the challenges of bonding our composites to the dentin surface. So, the components of composite resin materials we used and its manner of handling are critical factors to ensure it will work as it should be. One of the components of composite resins that works to unify its two different compositions, the organic and inorganic parts, is what we called coupling agents. Coupling agents acts as a chemical bond for the filler particle and the resin matrix to join together as one to disperse stress that’s brought upon on the restoration during the performance of tooth function like mastication. It is also a bifunctional molecule or what’s known as Organosilane that attaches the siloxane end bonds to hydroxyl groups found in fillers and the methacrylate end of the resin that polymerizes when exposed to light. The reason this is important in our practice is because we need to understand how it works, and why our composite resins cures as a whole considering it is made up of a solid and a liquid mass. Most of the coupling agents used by manufacturer is primarily made of silane. That’s why silane solution is the ideal material use in promoting adhesion in silica-based materials such as porcelains and used to restore damaged composite restorations.
Like always, I will discuss what’s this got to do with us? Although, it will be good to discuss the biochemistry on this topic, we might all yawn at the end of the day. What is important is to understand the relation of this material to our treatment. As I’ve mentioned, silane is one of the materials we used to achieve adhesion and it is ideal to silica-based and resin-based restorative materials, however, it is important to take note that it is not so in non-silica materials such as zirconia, metals and metal alloys.
The significant thing we have to take note is that when we are doing repairs on our restorations, the materials we have to use to bond one from another should be compatible, otherwise, it will not work.
For example, there’s one forum that a dentist suggested the use of Dycal base as a base and liner in a restoration with the final restorative material that will be used is composite resin. The restoration will not be durable and may cause certain problems later as Dycal, though evidence-base calcium hydroxide that works , is not compatible when the final restorative material to be used is composite resin. Since Dycal is a paste or cement type material and doesn’t contain any resin that will allow it to be bonded well on the composite resin. To make the explanation simpler, we can’t use ordinary paste to bond a ceramic or metal or wood or glass together. It can only be used to bond with paper. That’s the principle behind why we can’t use Dycal as a base when we use composite resin as our final restoration because the composite resin will end up just bonded on the lateral and occlusal walls of the tooth but not to the pulpal floor where the greatest force is absorbed during function.
Our Composite Resin Restoration in order to be durable must be bonded well to all tooth structure that it has a direct contact with.
It is also critical to take note that the shelf life for a single-bottle silane solution is relatively short. The solution will turn cloudy over time and cannot be used for adhesion. Two-bottle silane systems have been developed to offer a more stable system. One bottle contains an unhydrolyzed silane in ethanol and the other one contains an aqueous acetic acid solution. When mixed silane hydrolysis will take effect and necessary before use. Thus, for one bottle silane solution, it normally has an ideal shelf-life of 15 to 30 days in reality.
Therefore, what is the clinical significance of these coupling agents found in composite resins to our final restorations and bottom line to the quality of our treatment? It’s significance is that we need to prepare our cavity well in order for the bonding of our final restorations to the tooth will become effective. This is what we call surface conditioning. We need to condition our cavity preparation or the surface we will repair properly. The surface conditioning of restorative materials is an important preliminary step in clinical practice to modify surface properties for durable and hydrolytically stable adhesion. Here are the following surface conditioning we can do:
(1) Grit Blasting - The surface of materials such as metals, alloys and some ceramics is sand-blasted with alumina particles of 110 µm in size at a perpendicular distance of 10 mm under an air pressure of 380 kPa for ten to 15 seconds. This process is intended to increase the surface roughness of the materials. It also enhances micromechanical retention for bonding.
(2) Pyrochemical Silica Coating- Through the years, several silica-coating systems have been used in dental laboratories. For a brief time these are some materials used to achieved this: Silicoater Classical, Silicoater MD and Siloc (all Heraeus Kulzer) and PyrosilPen (SURA Instruments). In these systems, a tetraethoxysilane solution is injected into a flame and burned with butane in oxygen. The silane decomposes and forms reactive SiOx-C fragments, which are deposited on the substrate surface. A glass-like silica layer is thereby formed on the surface.
(3) Tribochemical Silica Coating- The Tribochemical Rocatec System introduced by 3M ESPE uses silica-coated alumina particles and was introduced in 1989. It is indicated for silica coating of ceramic and metal surfaces.
It enhances the adhesion of a silane coupling agent to a silica-coated material by forming a durable siloxane bond (Si-O-Si). This surface treatment also increases the surface roughness that provides micromechanical retention for resin bonding, that is, for the resin to penetrate pores on the surface.
(4) Hydrofluoric Acid Etching- Hydrofluoric acid is normally used to etch porcelain veneers and for intra-oral repair of fractured porcelain restorations before cementation. Low concentrations of 4 to 10 per cent hydrofluoric acid are used in clinical practice. When a porcelain surface is etched with hydrofluoric acid etching gel, the acid dissolves the glassy matrix of the porcelain. A microscopically porous and micro-retentive surface is thus produced and micromechanical interlocking for resin bonding is enhanced.
It is critical to take note that when one uses this as a method of surface conditioning and will be done inside the mouth of the patient, the dentist has to use a tissue protective material to prevent accidental tissue or gingival burning if rubber dam is not used.
The material should have the capability of neutralizing the hydrofluoric acid when it accidentally comes in contact with our patient’s gingiva. The most commonly used gingival barrier is sodium bicarbonate.
(5) Laser Surface Treatment- Laser stands for light amplification by stimulated emission of radiation and the technology was introduced in the 1950s. Er:YAG, Nd:YAG, and CO2 lasers are used in dentistry for soft-tissue surgery and hard-tissue treatment and surface treatment. Laser irradiation of a ceramic surface produces irregularities on the surface, which increase the surface roughness for mechanical retention. The main problem, however, of this surface treatment method is the formation of surface cracks owing to thermal effects of laser irradiation at high power settings. Therefore, appropriate laser settings for different ceramic surfaces is important to prevent formation of surface cracks.
(6) Selective Infiltration Etching- In this method, a thin layer of a glass conditioning agent is coated onto the zirconia surface and is then heated to above the glass transition temperature. The molten glass particles infiltrates between the surface grains. After this process, the specimens are allowed to cool at room temperature. The conditioning agent is then removed by applying hydrofluoric acid and rinsing it off. This creates a new retentive surface for resin–zirconia bonding.
(7) Nanostructured Alumina Coating- In this coating method, the zirconia is immersed in a suspension of aluminium nitride. Aluminium nitride undergoes hydrolysis to form boehmite, which is deposited onto the zirconia surface. A heat treatment at 900°C is carried out. Boehmite undergoes a phase transition to δ-alumina. Through this treatment, a micro-retentive surface area is created that may increase mechanical interlocking for resin bonding.
(8) Internal Coating with Porcelain- The zirconia surface is sand-blasted with alumina particles of 70 µm in size. Then, the surface is coated with high fusing porcelain, which is prepared by stirring the porcelain powder into an excess amount of distilled water. The porcelain is fired at a high temperature in a vacuum. After the firing process, the surface is sand blasted again. A silica-containing layer forms on the zirconia surface. This enhances adhesion with a silane coupling agent, that is, siloxane linkage formation.
(9) Chemical Vapour Deposition- In a chemical vapor deposition system, the zirconia surface is exposed to a vapour mixture of tetrachlorosilane and water. The silane hydrolyses and a SixOy seed layer is deposited as a coating on the surface. The thickness of the seed layer is controlled by deposition time. This silica seed layer provides the reactive sites for the silane coupling agent.
(10) Plasma Fluorination- In a plasma reactor, the zirconia surface is exposed to sulphur hexafluoride plasma. An oxyfluoride layer is formed on the surface. This layer may increase the reactivity of zirconia towards a silane coupling agent. However, the exact mechanism of the bonding formation between the zirconium oxyfluoride layer with silane is still unclear.
Surface condition is critical to allow bonding of 2 dissimilar materials. However, some of these procedures can’t be done within our clinic but used by laboratories. Thus, this is the reason why some of our final restorative material of choice is something we can’t or hard to repair clinically. So, here are the most common restorative materials we can probably repair on our own:
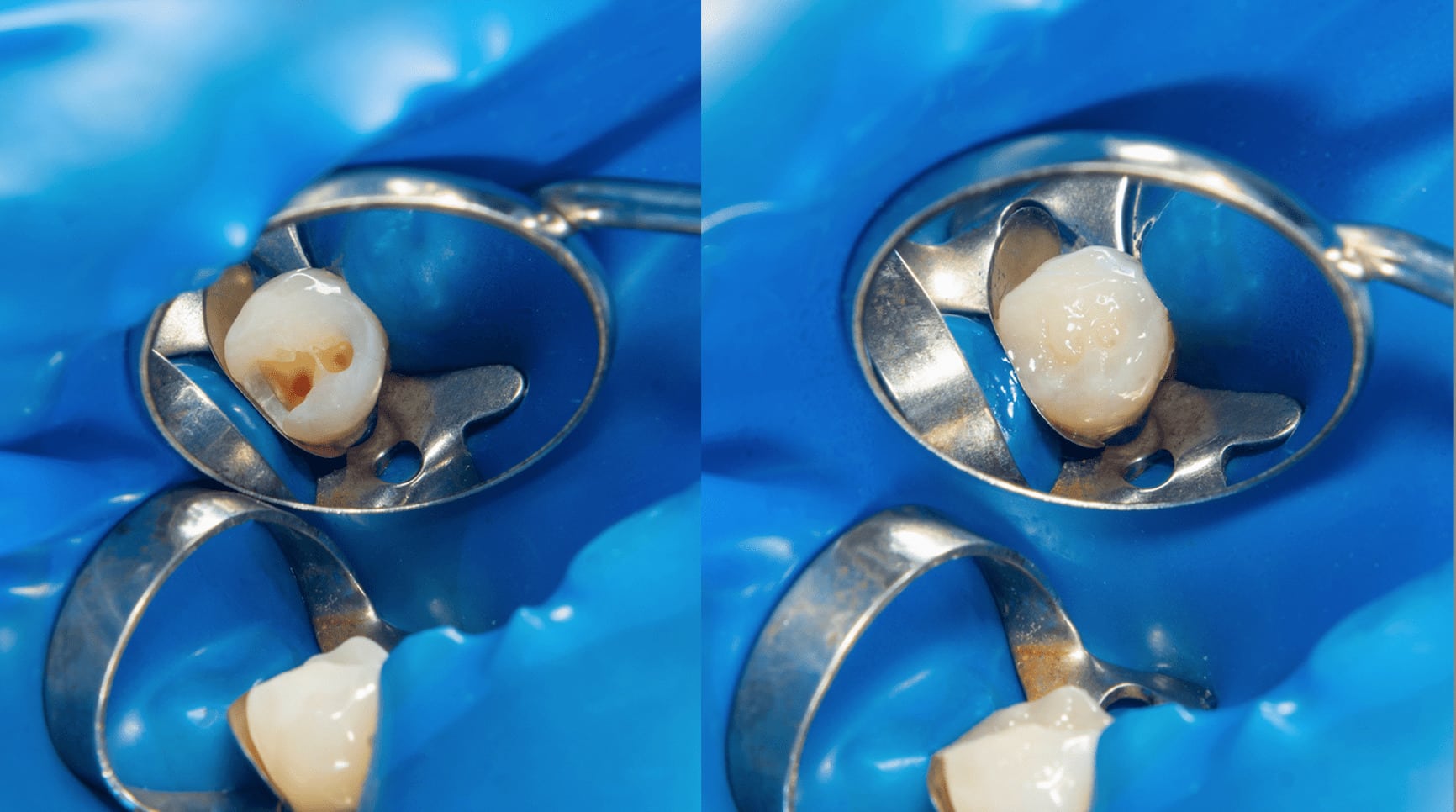
(1) Ceramic Restorations- Silane coupling agents are used in dental restoration, such as ceramic repairs of onlays, inlays, crowns and bridges. For most patients, repair is more economical and time-saving than the fabrication of new restorations, unless damage due to a fracture is beyond repair. The clinical procedure for repairing ceramic restoration usually involves the following steps: roughening the surface with diamond burs, sand-blasting the surface, acid etching, silanization and finally bonding to resin composite.
(2) Glass Fibre-Reinforced Composites
A relatively new group of dental biomaterials, the glass fibre-reinforced composites, is used in fixed partial dentures, removable prosthodontics, periodontal splints and retention splints. The adhesion between the glass fibre and resin composite is improved by adding a silane coupling agent. The silane forms siloxane linkages with the surface hydroxyl groups of glass fibre. The organo-functional groups of silane react with the functional group in the resin composite. Thus, the bonding strength is increased between resin composite and glass fibre.
(3) Resin Composite Filling Materials
Nowadays, dental resin composites are composed of a resin matrix that contains monomers and cross-linking monomers, as well as a free-radical initiator, an inhibitor, coloring pigments, filler materials such as barium glass, silica, apatite and a silane coupling agent. The latter enhances the bonding between the filler particles and the resin matrix. The filler particles added to the resin matrix also improve the physical and mechanical properties of the resin composite. Moreover, the addition of fillers reduces volume shrinkage after polymerization, and improves the aesthetic appearance and radiopacity.
(4) Titanium, Noble Metal and Base Metal Alloys
Titanium, noble metals and cobalt–chromium (base metal) alloys are commonly used for removable partial and complete dentures with a metal frame incorporated and metal–resin cement restorations. For these metal and metal alloys, surface conditioning by sand-blasting using silica-coated alumina particles produces a silica-coated layer on the surface. Application of a silane coupling agent to the silica-coated surfaces forms a durable siloxane linkage. This is followed by cementation.
Although, silanes are good at promoting adhesion between resin composites and dental restorative materials but there are some limitations to silane coupling agents.
The adhesion of silane coupling agents and non-silica-based restorative materials such as alumina, zirconia or metals is weaker than the silica coating of these materials. Therefore, a surface pre-treatment with silica coating is required so that durable bonds (siloxane bonds) are formed between silane and silica-coated restorative materials. For noble metals or noble metal alloys, thione or thiol-based coupling agents are used to promote adhesion. These coupling agents have different bonding mechanisms with various dental restorative materials.
Currently, other coupling agents (such as phosphate ester) are added to self-adhesive resin cements and adhesive primers, metal and alloy primers, and carboxylic acid primers used in dental restoration. Phosphate esters can bond directly to non-silica-based ceramics such as zirconia. It has been reported that using this phosphate ester can enhance the hydrolytic stability of bonding more than using silane coupling agents can.
The main problem of resin composites bonded to silica-coated restorative materials with the application of commercial silane coupling agents is the bond degradation over time under artificial ageing. In order to increase the hydrolytic stability of the bonding at the interfacial layer, novel surface treatments of restorative materials and the design of novel silane monomers can solve this problem. Silane coupling agents with long hydrocarbon chains are more hydrophobic than those with short hydrocarbon chains. The bonding at the interfacial layer is more resistance to thermal and water ageing. These two approaches could resolve the problem.
It could be said that silane coupling agents can fulfill the clinical requirements for dental restorations. Nowadays, a standard laboratory protocol for dental restorations entails surface conditioning of dental materials, silanization and cementation. The problem of hydrolytic stability of the siloxane linkage formed from silane coupling agents with resin composites and dental restorative materials is currently being addressed. Silane coupling agents do have a wide application in dentistry plays an important role in biomaterials science.
Therefore, knowing the components of your composite resin and why indeed the use of silane is necessary when we are doing repairs is critical. The use of any materials and how we bond them have to come to play to ensure that our restorative treatment will work and be durable for the sake of our patients.
[dvk_social_sharing] [et_bloom_inline optin_id="optin_1"]